Case Presentation
Case Presentation – May 2026
Primary Lung Adenocarcinoma in a Patient with Known Melanoma and Breast Cancer History
Written by: Michala Viano, Student, Cleveland Clinic School of Health Professions Cytology Program, Cleveland, Ohio
Patient Age: 81-year-old female
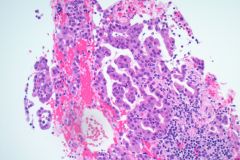
Specimen Type: FNA Station 4L, 4R Lymph Nodes, Modified Romanowsky smears, Papanicolaou smears, Thin Prep® Non-Gyn slide, H&E Cellient® Cell Block
Patient History: Breast Ductal Carcinoma in Situ (DCIS) with lumpectomy and adjuvant radiation treatment (1997), melanoma of the midback (2005), incidental right lung mass consistent with Lung Adenocarcinoma with evidence of intracranial metastasis (2025)
Cytologic Diagnosis: Positive for Malignancy, Adenocarcinoma
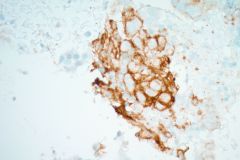
Biopsy/Pathologic Diagnosis: Corresponding Surgical Pathology H&E Slides of Core Biopsy of 4R Lymph Node, Positive for Malignancy, Adenocarcinoma with malignant cells staining positive for TTF-1 and negative for GATA3 and SOX10. PD-L1 immunohistochemical staining is positive.
Case provided by: Cleveland Clinic, Cleveland, Ohio
Primary Lung Adenocarcinoma in a Patient with Known Melanoma and Breast Cancer History
Etiology:
Adenocarcinoma is the most common type of lung cancer and the most frequently diagnosed lung malignancy in nonsmokers and females.1,2 It arises from glandular epithelial cells in the peripheral lung, most commonly type II pneumocytes and bronchial epithelial cells.1,7 While cigarette smoking is the strongest environmental risk factor for lung cancer overall, lung adenocarcinoma can still develop in patients without a smoking history.2,3 Additional contributing factors include long-term environmental exposures such as air pollution, occupational inhalants, and chronic lung injury or irritation, which result in repeated epithelial damage and repair over time.2,3 With repeated repair, the epithelium becomes more prone to replication errors and DNA mutations that allow cells to proliferate uncontrollably.5 Common alterations include mutations in EGFR and KRAS, rearrangements involving ALK or ROS1, and alterations in TP53.⁶ The accumulation of these genetic changes ultimately leads to malignant transformation of the lung epithelium.5 It is also important to note that the lung is an extremely common site for metastatic disease due to the ease of tumor cells depositing while traveling through the bloodstream. The most common metastatic cancers to appear in the lung are breast and colorectal adenocarcinoma, as well as melanoma.8
Clinical Features:
Patients with lung adenocarcinoma may initially be asymptomatic, particularly in the early stages when the tumor is small and located in the peripheral lung and airway obstruction is minimal.1,2 As it progresses, patients commonly develop respiratory symptoms including persistent or chronic cough, worsening shortness of breath (dyspnea), chest pain, fatigue, and unexplained weight loss, with some patients producing increased sputum, developing a pleural effusion, or experiencing hemoptysis (coughing up blood).3 In advanced stages when metastasis is likely, the patient may begin experiencing neurological symptoms, bone pain, or systemic weakness, and the brain, bones, liver, adrenal glands, and pleura should be monitored as they are common lung adenocarcinoma metastatic sites.1,2 Imaging is almost always necessary, and many lung nodules are incidental findings while a patient is being examined or treated for an unrelated condition.1,2
Treatment and Prognosis:
Treatment for lung adenocarcinoma depends mainly on the stage at diagnosis and whether DNA mutations are present.1,2 In early-stage disease confined to the lung, surgical resection is often the primary treatment and may involve a wedge resection, lobectomy, or removal of an entire lung depending on the tumor size and location.1 Adjuvant chemotherapy or radiation therapy may be added after surgery based on lymph node involvement and other high-risk tumor features. 1,2 In advanced or metastatic stages, when the disease is no longer confined to the lung, systemic therapy becomes the primary treatment and may include platinum-based chemotherapy, targeted therapy directed at specific mutations such as EGFR or ALK, immunotherapy, or a combination of these treatments. 2,6 Immunotherapy selection is often guided by PD-L1 expression, which is evaluated using immunohistochemical staining. In this case, the tumor shows strong PD-L1 expression, supporting the use of immunotherapy as a treatment option. 12 Prognosis varies widely depending on the stage at diagnosis.4 Patients with localized lung cancer have an estimated 5-year survival rate of about 60–70%, those with lymph node involvement have survival rates closer to 30–40%, and patients with distant metastatic disease have a 5-year survival rate below 10%.4 Outcomes have improved in recent years due to targeted therapies, with some mutation-positive patients experiencing longer disease control and improved survival. 2,6 The main goals of treatment are to prolong survival, slow disease progression, and maintain quality of life.1
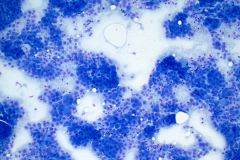
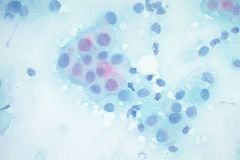
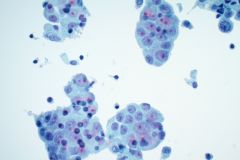
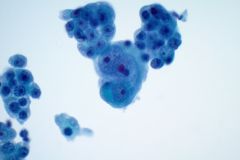
Cytology:
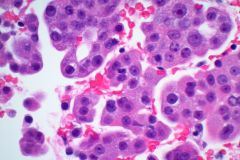
When reviewing the slides cytologically, there is moderate to high cellularity, with malignant cells present in three-dimensional cohesive groups as well as scattered single cells.1,7 Compared to the background population, these cells appear enlarged and demonstrate increased nuclear-to-cytoplasmic ratios, irregular nuclear borders and contours, and prominent nucleoli with variable chromatin patterns.1 Nuclear pleomorphism is present, and occasional multinucleated tumor cells can be identified.1 The cytoplasm is moderate in amount and appears vacuolated due to mucin production, which reflects glandular differentiation with extracellular mucin and necrotic debris present in the background.1,2 Overall, these cytologic findings are characteristic of an epithelial glandular cell malignancy. Because adenocarcinoma shares overlapping cytologic features with other poorly differentiated tumors, IHC stains such as TTF-1 and Napsin A are used to help confirm pulmonary origin.1,7
Differential Diagnosis:
Given the patient’s prior clinical history of breast cancer, metastatic breast adenocarcinoma was an important differential diagnosis to consider. Cytologically, metastatic breast adenocarcinoma can present with cohesive clusters of epithelial cells, enlarged nuclei, and prominent nucleoli, which may overlap with the appearance of lung adenocarcinoma.9 Because these tumors may share similar glandular morphology, IHC staining is often necessary to determine the primary site. Breast adenocarcinoma typically demonstrates positivity for markers such as estrogen receptor (ER), progesterone receptor (PR), GATA3, and mammaglobin.9
Metastatic melanoma was also an important consideration given the patient’s history. Melanoma may show pleomorphic cells with prominent nucleoli, abundant cytoplasm, and variable cellular cohesion, which can sometimes resemble poorly differentiated carcinoma on cytologic preparations.10 Because melanoma demonstrates wide morphologic variability, immunohistochemical stains are essential for diagnosis. Melanoma typically shows positivity for markers including S100, SOX10, HMB-45, and Melan-A, which help distinguish it from epithelial malignancies.10
Another important differential diagnosis is metastatic colorectal adenocarcinoma, as colorectal tumors commonly metastasize and may demonstrate gland-forming epithelial cells with mucin production that can resemble pulmonary adenocarcinoma.11 IHC stains such as CK7, CK20, and CDX2 are commonly positive in colorectal adenocarcinoma. 1,11 Correlation of morphologic findings with IHC staining is essential in distinguishing between these potential primary sites.
References:
- Myers DJ, Wallen JM. Lung adenocarcinoma. Nih.gov. Published June 12, 2023. https://www.ncbi.nlm.nih.gov/books/NBK519578/
- Lemjabbar-Alaoui H, Hassan OU, Yang YW, Buchanan P. Lung cancer: Biology and treatment options. Biochimica et Biophysica Acta (BBA) – Reviews on Cancer. 2015;1856(2):189-210. doi: https://doi.org/10.1016/j.bbcan.2015.08.002
- American Cancer Society. Lung Cancer Signs & Symptoms | Common Symptoms of Lung Cancer. www.cancer.org. Published January 29, 2024. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Lung Cancer Survival Rates | 5-Year Survival Rates for Lung Cancer. www.cancer.org. Published January 29, 2024. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/survival-rates.html
- Cooper GM. The Development and Causes of Cancer. Nih.gov. Published 2000. https://www.ncbi.nlm.nih.gov/books/NBK9963/
- Pao W, Girard N. New driver mutations in non-small-cell lung cancer. The Lancet Oncology. 2011;12(2):175-180. Doi: https://doi.org/10.1016/s1470-2045(10)70087-5
- Travis WD, Brambilla E, Nicholson AG, et al. The 2015 World Health Organization Classification of Lung Tumors. Journal of Thoracic Oncology. 2015;10(9):1243-1260. doi: https://doi.org/10.1097/jto.0000000000000630
- Jamil A, Kasi A. Lung Metastasis. PubMed. Published 2022. https://pubmed.ncbi.nlm.nih.gov/31971751/
- Harbeck N, Gnant M. Breast cancer. The Lancet. 2017;389(10074):1134-1150. doi: https://doi.org/10.1016/s0140-6736(16)31891-8
- Heistein J, Acharya U. Cancer, Malignant Melanoma. PubMed. Published 2023. https://www.ncbi.nlm.nih.gov/books/NBK470409/
- Bayrak R, Haltas H, Yenidunya S. The value of CDX2 and cytokeratins 7 and 20 expression in differentiating colorectal adenocarcinomas from extraintestinal gastrointestinal adenocarcinomas: cytokeratin 7-/20+ phenotype is more specific than CDX2 antibody. Diagnostic Pathology. 2012;7(1):9. doi: https://doi.org/10.1186/1746-1596-7-9
- Schoenfeld AJ, Rizvi H, Chaitanya Bandlamudi, et al. Clinical and molecular correlates of PD-L1 expression in patients with lung adenocarcinomas. Annals of Oncology. 2020;31(5):599-608. doi: https://doi.org/10.1016/j.annonc.2020.01.065