Case Presentation
Case Presentation – August 2023
Metastatic Ductal Breast Carcinoma to the Lung
Written by: Jenna Kerns, Student, Cleveland Clinic School of Cytotechnology, Cleveland, Ohio
Patient Age: 45 year old female
Specimen Type: FNA of Station 7 Lymph node, Diff-Quik and Papanicolaou Stained smears, ThinPrep® Non-Gyn cytology, Formalin-Fixed Cell Block
Patient History: Invasive Ductal Breast Carcinoma (T2N1M0 ER+PR+, Her2neu-negative right breast) w/ adjuvant radiation (2008)
Cytologic Diagnosis: Positive for Malignant Cells, Metastatic Carcinoma of Breast Origin. The malignant cells are positive for GATA3, AE1/AE3 and estrogen receptor. P40, TTF-1 and Napsin A are negative.
Biopsy / Pathologic Diagnosis: Metastatic Adenocarcinoma consistent with patient’s known history of a breast cancer primary. Immunohistochemical stains including a GATA3 and TTF-1 were used in evaluation of this material. The neoplastic cells show strong and diffuse expression of GATA3 and are negative for expression of TTF-1, supporting a metastasis from the patient’s known breast primary. Immunohistochemical stains for estrogen receptor and progesterone receptor were both positive and HER2 was negative.
Case provided by: Cleveland Clinic, Cleveland, Ohio
Metastatic Ductal Breast Carcinoma in the Lung
Etiology:
Breast cancer is the leading type of cancer affecting women in the United States.1,2 It was estimated that in 2022 there would be 290,560 new cases of breast cancer (287,850 female and 2,710 male) and 43,780 deaths from breast cancer (43,250 female and 530 male) in the United States.2,3 Unfortunately, breast cancer is a heterogeneous disease and has many root causes; some of which can and cannot be avoided. It also consists of many different types of tumors.4 Of the different types of malignant breast tumors, invasive ductal breast carcinoma (IDC) is the most commonly diagnosed type and accounts for 40-75% of breast malignancies.1 Some of the biggest root causes for developing IDC are being a carrier of the genetically mutated BReast CAncer genes 1 and 2, (BRCA1 and BRCA2), having too many copies of the Human Epidermal growth factor Receptor 2 (HER2) gene, and having Estrogen Receptors (ER) and Progesterone Receptors (PR).5,6 In addition to these genetic causes, other factors such as being female, aging, having an inherited risk from first-degree relatives, and increased breast density increase the risk of developing IDC. Menopause hormonal therapy (MHT), combination hormone therapy, estrogen therapy, ionizing radiation, obesity, drinking alcohol, and smoking can also contribute to the development of IDC. 2,5,7 Invasive ductal breast carcinoma is most commonly diagnosed in white women ages 55 and older.5,8 The patient in our case study has a family history of breast cancer in her mother, two aunts and a maternal cousin, is negative for the BRCA1 and BRCA2 gene and is a never smoker.
Clinical Features:
Ductal breast carcinoma originates from the cells that line the milk ducts of the breast. This cancer is considered invasive once it spreads to the surrounding normal tissue outside of the ducts.2,8,9 If left unattended or not treated properly, ductal carcinoma can continue to spread in the surrounding tissue, involving the lymph nodes and blood vessels. From here, IDC has the potential to metastasize to distant organs of the body, such as the liver, lungs, bone and brain.8,10,11 One of the most common clinical symptoms of IDC is a palpable lump in the breast, which can be found during a self-breast exam or during an office visit with a doctor. Other symptoms such as thickening or redness of the breast, dimpling of the skin of the breast or nipple, abnormal swelling and change in shape of the breast, nipple discharge that is not breast milk, continuous localized breast pain, and lumps in the underarm are also characteristic to IDC. Additionally, IDC can also be asymptomatic in patients.1,2,8 Mammograms are important and serve as a great screening method, catching suspicious calcifications or small non-palpable lumps within the breast.1,2,8 When IDC metastasizes to a patient’s lungs, the patient can experience clinical symptoms such as pain in the lung, difficulty breathing and shortness of breath, fatigue, hemoptysis, and unintentional weight loss.11 In our case study, the patient presented with a right pleural effusion with associated atelectasis and an enlarged 2.2 x 1.5cm subcarinal lymph node with max SUV of 3.3 concerning for recurrent disease. She also noted in her office visit that she had localized right breast pain and pain in the chest before receiving a CT and PET scan.
Treatment and Prognosis:
The prognosis of a patient with breast cancer is based on the original stage at diagnosis and the molecular subtype.2,6 A patient with ductal carcinoma in situ (DCIS), a cancer that has not spread past the basement membrane of the ducts, has a 5-year survival rate of 99.1%. When ductal breast carcinoma becomes invasive and infiltrates the lymph nodes and blood vessels, the patient’s 5-year survival rate decreases slightly to 86.1%. If ductal breast carcinoma continues to spread and metastasizes to distant organs of the body, the patient’s 5-year survival rate decreases greatly to 30.0%.2,11 The four different molecular subtypes are based on a combination of whether a patient is positive or negative for the following: too many copies of the HER2 gene, estrogen receptors, and progesterone receptors.6,12 The first molecular subtype, Luminal A, is the most common and accounts for 40% of breast cancers. Here the patient is ER (+) and/or PR (+), HER2 (-). Patients with this subtype have the least aggressive form of breast cancer and have a 5-year survival rate of 94.4%. The second most common molecular subtype is Luminal B and it accounts for 20% of breast cancers. A patient that has the Luminal B molecular subtype will be ER (+) and/or PR (+), HER2 (+). This is more aggressive than Luminal A, and the 5-year survival rate is 90.7%. The next molecular subtype, HER2 positive, accounts for 10-15% of breast cancers. This is the rarest molecular subtype and patients are ER (-), PR (-), HER2 (+). This is also a more aggressive form of breast cancer and the 5-year survival rate is 84.8%. The last molecular subtype, Triple Negative/ Basal-Like, is the most aggressive form of breast cancer and has a 5-year survival rate of 77.1%. It accounts for 10-15% of breast cancers and the patient is ER (-), PR (-), HER2 (-).6,10,12 In the current case presentation, the patient has the Luminal A molecular subtype of metastatic ductal breast carcinoma (MDC).
There is a wide variety of treatment options for patients with breast cancer. Treatments for MDC include hormone therapy, targeted therapy, chemotherapy, immunotherapy, surgery, and radiation therapy.5,8,11,13 Hormone therapy works well in patients who have MDC and are ER (+) and/ or PR (+). This type of treatment consists of two commonly used therapies, tamoxifen and aromatase inhibitors. Targeted therapies work best in patients who have MDC and are ER (+) and/ or PR (+), but have not responded well to other treatments, or patients who have MDC and are HER2 (+). Examples of these treatments are mTOR inhibitors, CDK4/6 inhibitors, and cyclin-dependent kinase inhibitors. Patients who have MDC and are ER (-) and PR (-) usually receive chemotherapy. Radiation therapy can be used to relieve the symptoms and improve the quality of life for patients with MDC.6,11,13 In the current case presentation, the patient was treated with leuprolide, targeted therapies abemaciclib (a type of cyclin-dependent kinase inhibitor) and CDK4/6 inhibitors, and the hormone therapy letrozole (a type of aromatase inhibitor). Before the breast cancer metastasized to her lungs, she was treated with a right partial mastectomy, radiation therapy and tamoxifen.
Cytology:
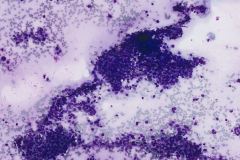
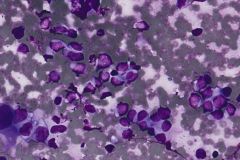
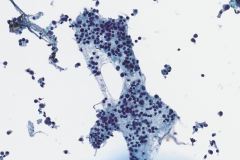
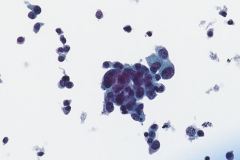
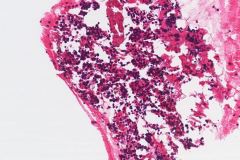
Ductal breast carcinoma specimens are highly cellular and consist of a variety of cell arrangements. The cells will commonly be seen as poorly cohesive clusters, some isolated single cells, and rare sheets of cells. These specimens will have enlarged and hyperchromatic nuclei that are eccentrically placed and often protruding from the cytoplasm. The chromatin has a bland appearance, ranging from finely to coarsely granular, with prominent, irregularly shaped nucleoli. Myoepithelial cells sitting on top of or lining the ductal breast cells are helpful in determining if the specimen is benign and should not be present in IDC specimens. The background of IDC is usually clean, but inflammation, blood, and granular debris may be observed.1,14 Immunohistochemistry (IHC) markers that can be used to stain for ductal breast carcinoma are Ki-67, p63, CK5/6, ER, Mammaglobin, GCDFP-15, E-Cadherin and GATA3. Double staining for cytokeratin, smooth muscle myosin heavy chain, Calponin, and/or p63 is helpful for determining if myoepithelial cells are present in the specimen.1,15 In the current case study, the patient’s specimen presented with marked cellularity consisting of a population of both 3-dimensional clusters of cells and many dyshesive single cells. In the smaller 3-dimensional clusters, some of the cells can be seen forming glandular structures and some look as if they are molded together. The nuclei are enlarged, eccentrically placed, and have a mix of both smooth and irregular nuclear contours. The cells all have high N/C ratios and there is a moderate amount of anisonucleosis. The chromatin has a predominantly hyperchromatic appearance, is finely granular and large prominent nucleoli can be observed. The cytoplasm is scant, has well-defined cytoplasmic borders, and is finely vacuolated. Mucinous vacuoles in the cytoplasm can be seen in the larger 3-dimensional clusters of cells. Crush artifact and necrosis can be observed in the background.
Differential Diagnosis:
The cytologic differential diagnoses for IDC include primary lung adenocarcinoma, small cell carcinoma, and basaloid squamous cell carcinoma. IHC stains are useful ancillary tests that aid in determining a diagnosis.
- Primary Lung Adenocarcinoma: Adenocarcinoma is the most common subtype of primary cancer in the lung. It is commonly seen in women, young men, and non-smokers.1 Primary Lung Adenocarcinoma specimens are highly cellular specimens with 3-dimensional clusters that can take on an acinar or papillary appearance. The nuclei are enlarged, eccentrically placed, have smooth nuclear contours, and contain large prominent nucleoli. The chromatin has a finely granular texture which gives the nuclei a bland appearance. The cytoplasm is abundant, has well-defined borders and contains large or fine mucinous vacuoles. Cells from primary lung adenocarcinoma have high N/C ratios, but no molding is observed.1,14 IHC markers that can be used to stain for primary lung adenocarcinoma are TTF-1, p63 or p40, CK5/6, Napsin A, and Mucicarmine.1 In the current case study, the patient’s specimen presented with marked cellularity and consisted of a population of both 3-dimensional clusters of cells as well as dyshesive single cells. The single cells are not characteristic of primary lung adenocarcinoma, but they have similar chromatin patterns and look like the cells in the small clusters. Crush artifact can also be seen in the background, which is not usually seen in primary lung adenocarcinoma.
- Small Cell Carcinoma: Small cell carcinoma is a type of neuroendocrine tumor that accounts for 20-25% of all primary lung cancers. This type of cancer is commonly seen in male smokers and usually has a poor prognosis.1 Small cell carcinoma specimens are moderately cellular with cells arranged in a dispersed, single cell population. Cells from small cell carcinoma have scant to no cytoplasm and exhibit a coarse chromatin pattern. Moderate pleomorphism can also be observed. Nuclear molding, crush artifact, and inconspicuous nucleoli are characteristic features of small cell carcinoma. In the background, marked necrosis, abundant mitotic figures and apoptotic bodies can also be observed. Paranuclear blue blobs may be seen amongst the small cells.1,14 IHC markers that can be used to stain for small cell carcinoma are Chromogranin, Synaptophysin, INSM1, AE1/3, and CD56.1 The smaller 3-dimensional clusters of cells from the patient of the current case study look as if they are molded together, which might suggest small cell carcinoma, but it is not true molding. The cells also have eccentrically placed nuclei with small amounts of cytoplasm, which is not characteristic of small cell carcinoma.
- Basaloid Squamous Cell Carcinoma: Basaloid squamous cell carcinoma is a highly aggressive form of squamous cell carcinoma. Basaloid squamous cell carcinoma specimens present with predominantly cohesive tissue fragments and single keratinized cells. Nuclear atypia such as molding, inconspicuous nucleoli, and high N/C ratios are characteristic to basaloid squamous cell carcinoma. The nuclei range in size from small to medium and have finely granular chromatin.16 IHC markers that can be used to stain for basaloid squamous cell carcinoma are CK5/6 and p63. Basaloid squamous cell carcinoma also stains negative for TTF-1, synaptophysin, and chromogranin.16 In the current case study, large prominent nucleoli and finely vacuolated cytoplasm can be observed, which are not normally present in basaloid squamous cell carcinoma. Single keratinized cells, which are characteristic of basaloid squamous cell carcinoma, are not present.
References:
- Cibas E. S., and Ducatman B. S. Cytology: Diagnostic Principles and Clinical Correlates: Fifth Edition. 2021. Elsevier Inc.
- Breast Cancer-Health Patient Version. National Cancer Institute. https://www.cancer.gov/types/breast/hp. Accessed February 19, 2023.
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72(1):7-33. doi:10.3322/caac.21708
- Zhao H. The prognosis of invasive ductal carcinoma, lobular carcinoma and mixed ductal and lobular carcinoma according to molecular subtypes of the breast. Breast Cancer. 2021;28(1):187-195. doi:10.1007/s12282-020-01146-4
- Breast Cancer-Health Professional Version. National Cancer Institute. https://www.cancer.gov/types/breast/hp. Accessed February 19, 2023.
- Breast cancer: Different types, different treatments. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/breast-cancer/in-depth/breast-cancer/art-20045654. Published December 6, 2022. Accessed February 22, 2023.
- Jordahl KM, Malone KE, Baglia ML, et al. Alcohol consumption, smoking, and invasive breast cancer risk after ductal carcinoma in situ. Breast Cancer Res Treat. 2022;193(2):477-484. doi:10.1007/s10549-022-06573-9
- Wright P. Invasive Ductal Carcinoma (IDC). John Hopkins Medicine. https://www.hopkinsmedicine.org/health/conditions-and-diseases/breast-cancer/invasive-ductal-carcinoma-idc#:~:text=The%20risk%20of%20getting%20invasive,IDC%20can%20affect%20men. Published January 21, 2022. Accessed February 19, 2023.
- NCI Dictionary of Cancer terms. National Cancer Institute. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/invasive-ductal-carcinoma. Accessed February 19, 2023.
- Breast cancer – metastatic – introduction. Cancer.Net. https://www.cancer.net/cancer-types/breast- cancer- metastatic/introduction#:~:text=It%20most%20often%20spreads%20to,breast%20cancer%2C%20not%20lung%20cancer. Published June 3, 2022. Accessed February 21, 2023.
- Eske J. What Happens When Breast Cancer Metastasizes in the Lung? Medical News Today. January 2019. https://www.medicalnewstoday.com/articles/324304. Accessed February 22, 2023.
- Female breast cancer subtypes – cancer stat facts. SEER. https://seer.cancer.gov/statfacts/html/breast-subtypes.html#:~:text=Survival%20Statistics&text=The%20best%20survival%20pattern%20was,subtype%20had%20the%20worst%20survival. Accessed March 2, 2023.
- Trayes KP, Cokenakes SEH. Breast Cancer Treatment. Am Fam Physician. 2021;104(2):171-178.
- Keebler C. M., and Somrak T. M. The Manual of Cytotechnology: Seventh Edition. 1993. The American Society of Clinical Pathologists
- Bonacho T, Rodrigues F, Liberal J. Immunohistochemistry for diagnosis and prognosis of breast cancer: a review. Biotech Histochem. 2020;95(2):71-91. doi:10.1080/10520295.2019.1651901
- Marks RA, Cramer HM, Wu HH. Fine-needle aspiration cytology of basaloid squamous cell carcinoma and small cell carcinoma-a comparison study. Diagn Cytopathol. 2013;41(1):81-84. doi:10.1002/dc.21853